The Breakthrough
James M. Murphy and his team have their experimental result published on Immunity in 2013. On this page, we have summarized the article's details and will give you an overview of their research.
Objectives
It is important that revealing the structure of the MLKL protein can help to define the role and mechanism of the protein's action. Thus, the team was trying to solve the structure of the MLKL protein by X-ray Crystalography. MLKL is a component of the necrosome - a multiprotein complex which triggers tumor necrosis factor-induced cell death (necroptosis).
How ?
To determine the crystal structure of the MLKL protein, the team has concentrated full-length MLKL proteins to 5mg/ml for protein crystallization and subjected the sample to robotic crystal trials. The structure of the protein was determined by X-ray diffaction at a resolution of 2.6 Å.
Since MLKL protein is a major component of necrosome, the cells cannot enter necrotopsis programmed cell death if the protein is absent. Therefore, The team had generated a group of MLKL-deficient mice which were kept in stress-free conditions. The rodents were healthy and fertile, so all the cells taken from the mice bodies were resistant to TNF-induced necroptotic cell death. The team delivered the MLKL-deficient cell from the mice, and reconstituted the cells with some MLKL expression constructs to test the consequences of introducing structure-guided MLKL mutations. This procedures were performed to investigate the role and mechanism of the MLKL protein.
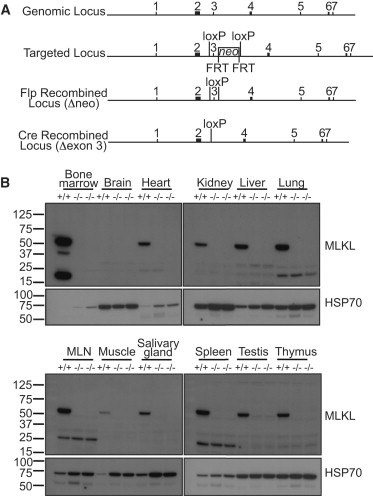
The gel in Figure 1 below shows that MLKL protein was absent in all tissues of homozygous mutant mice generated by the team.
Since MLKL protein is a major component of necrosome, the cells cannot enter necrotopsis programmed cell death if the protein is absent. Therefore, The team had generated a group of MLKL-deficient mice which were kept in stress-free conditions. The rodents were healthy and fertile, so all the cells taken from the mice bodies were resistant to TNF-induced necroptotic cell death. The team delivered the MLKL-deficient cell from the mice, and reconstituted the cells with some MLKL expression constructs to test the consequences of introducing structure-guided MLKL mutations. This procedures were performed to investigate the role and mechanism of the MLKL protein.
The gel in Figure 1 below shows that MLKL protein was absent in all tissues of homozygous mutant mice generated by the team.
Results and Findings
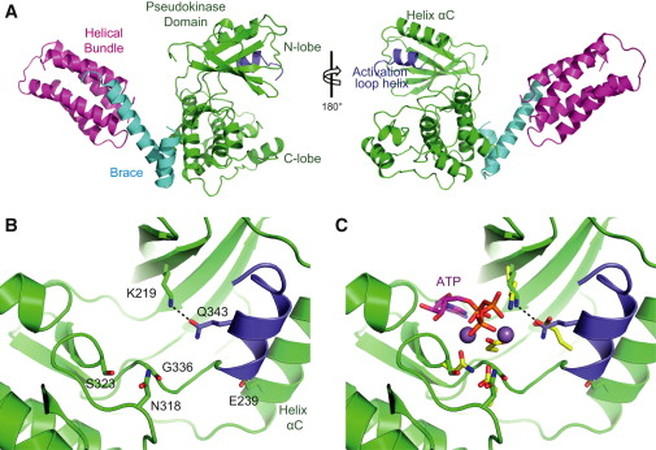
The team had found that a full length MLKL protein contains a N-terminal four-helix bundle which is tethered to the C-terminal pseudokinase domain. The result shows that the N-terminal domain comprises of a four-helix bundle followed by a two helix linker that connecting the whole bundle to the pseudokinase domain. A more detail looks of the whole structure shown in Figure 1 below.
Meanwhile, the team found that the dermal fibroblasts, embryonic fibroblasts , and bone-marrow-derived macrophages of the homozygous mutant mice (Mlkl−/−) were resistant to necrotopsis, in clear contrast to their wild-type counterparts. In addtion, a reconstitution of full-length MLKL to the Mlkl−/− mice has restored the sensitivity of bone-marrow-derived macrophages ((Mlkl−/−) ) to TNF-induced necrotopsis.This finding strongly supports that MLKL protein is required for necroptosis
Some other features were concluded by the team:
Some other features were concluded by the team:
- MLKL psuedoactive site mutants can induce necroptosis in the absence of exogenous stimuli
- Necroptosis induced by MLKL psuedoactive site mutants is independent to RIPK3 activiy
- PAGAM5, a mitochodrial protein is dispensable for RIPK3-MLKL-mediated necroptosis
The Original Article
James M. Murphy, Peter E. Czabotar, Joanne M. Hildebrand, Isabelle S. Lucet, Jian-Guo Zhang, Silvia Alvarez-Diaz, Rowena Lewis, Najoua Lalaoui, Donald Metcalf, Andrew I. Webb, Samuel N. Young, Leila N. Varghese, Gillian M. Tannahill, Esme C. Hatchell, Ian J. Majewski, Toru Okamoto, Renwick C.J. Dobson, Douglas J. Hilton, Jeffrey J. Babon, Nicos A. Nicola, Andreas Strasser, John Silke, Warren S. Alexander. The Pseudokinase MLKL Mediates Necroptosis via a Molecular Switch Mechanism. Immunity, 2013
http://www.sciencedirect.com/science/article/pii/S1074761313003488
http://www.sciencedirect.com/science/article/pii/S1074761313003488