Necroptosis
Receptor interacting protein (RIP) kinases constitute a family of seven members, all of which have a kinase domain. They are essential regulators of cell survival and death. Definitely, RIP1 kinase activation is vital as an upstream regulator of necroptotic death pathway.
There are two kinase, which are receptor interaction protein 1 & 3 (RiP1), (RiP3), play a key role in regulate of necroptosis. Apoptosis and necrosis can stimulated by RiP1; whereas, RiP3 seems to acts on necroptosis only. There will be a functional signalling of a amyloid complex formed from RiP1 and RiP3 in RHIM-RHIM domain interaction. C-terminal RHIM domain is included in these kinases (Chen , 2012)
The complex biochemistry of necroptosis can be scrutinized in the video below.
Zhae (2013) states that necrosome complex mediates the tumor necrosis factor (TNF) –induced necroptosis. During induction of necroptosis Rip3 associates and phosphorylates (MLKL), but this phosphorylation is needed for Rip3 dependent necroptosis.
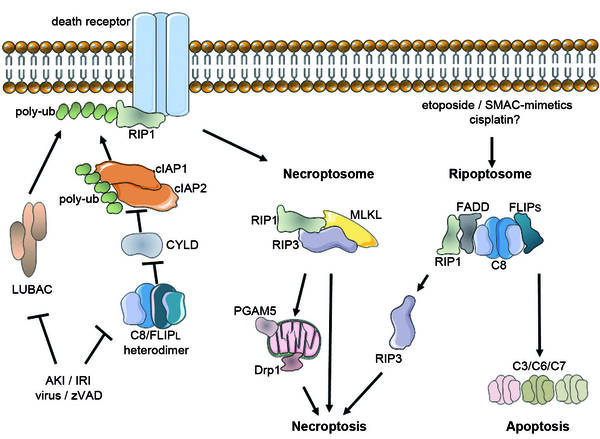
In unprocessed cells, death receptor ligation marks in activation of the apoptosis cascade heterodimer that inactivates crucial actors of necroptosis like Cylindromatosis (CYLD), which acts as tumor suppressor, then apparently leads to the cleavage of RIP1 and RIP3. RIP1 will bind to RIP3 and trigger the downstream cascade of necroptosis, a typical example for immunogenic cell death plus programmed necrosis. The RIP1-RIP3-interaction and consequences in chains of phosphorylation events, possibly including an unidentified kinase. This results from the recruitment of MLKL and the assembly of the supramolecular construction called the necroptosome. This assembly is essential for further transduction of the necroptosis signal into mitochondria, but the downstream partners of RIP3 and the global dependence on mitochondria are presently seriously debated (Zhae, 2013).
Cell death induced by the activation of the death receptor may be accomplished through alternative death pathways or necroptosis.
Necroptosis is also implicated in developmental processes (Kaczmarek, 2013). When FADD or caspase 8 is defficient in mice the embryo is spontaneously aborted. The stage of abortion is that where hematopoietic development occurs, this may be related to the way necroptosis regulates activated T-cells. Necroptosis regulates T-cells in periphery tissues seemingly by eliminating excessive or abnormal lymphocytes.
The regulation of activated T-cells is not fully understood, however defects in the necroptosis pathway lead to T-cell proliferative diseases. The lymphocytes produced from these disorders are very abnormal, this indicates that necroptosis has greater functions in developmental processes and is not only an alternative method of compensation when apoptosis is dysfunctional.
Many Damage Associated Molecular Patterns (DMAPs) have been identified to trigger the Necroptosis pathway, however much research is still being done to identify those that are capable of triggering or sensitising the Necrosome to activation without due cause. DAMPs as well as deffects in the Necrosome proteins are likely the causes of many inflamatory disorders & need further investigation.
Some useful links
Here are some website/articles we believe that they are useful for further understanding the biochemistry signalling pathway of Necroptosis. We strongly recommand you to have a look on them.